Le blog Sparky

par Ethan Wilson, M.Sc., B.Sc
•
19 février 2026
Spirometry is one of the most widely used pulmonary function tests and is often considered the gold standard in respiratory diagnostics (1–3). While the test itself is relatively simple in concept, obtaining reliable and high-quality results requires adherence to guidelines established in the joint statement by the American Thoracic Society (ATS) and European Respiratory Society (ERS). Preschool children can successfully perform spirometry—but they often have difficulty meeting standard ATS/ERS criteria (2, 4). Understanding best practices, age-appropriate modifications, and the differences between in-clinic and at-home spirometry is key to interpreting results correctly. Why Spirometry Is Challenging in Preschoolers Spirometry requires coordination, effort, and sustained forced expiration—all of which can be difficult for young children. Preschoolers may (2): Struggle to maintain a forced expiration for more than one second Have short attention spans and are easily distracted Require encouragement and engagement Cannot perform the maneuvers required for some pulmonary function tests Equipment for this age group is often designed for older and larger individuals Because of these developmental factors, adaptations are often necessary to obtain meaningful results. Best Practice Patient Guidelines for In-Clinic Spirometry in Preschool Children According to the joint statement from the ATS and ERS, as well as the Canadian Thoracic Society (CTS) the following guidelines should be followed for proper validity and repeatability of spirometry in children (2, 5, 6): NOTE that technical spirometry guidelines are not covered in this article and readers should refer to the official joint statement of the ATS/ERS 1. Patient Positioning Proper positioning is essential for safety and optimal performance: Sit upright Keep feet flat on the floor with legs uncrossed Loosen tight-fitting clothing Use a chair with armrests to prevent falls if lightheaded Even small deviations in posture can influence results, so positioning should always be standardized and documented. 2. Important considerations for Children: The child should receive training and familiarization with the equipment A bright room and a happy, pleasant environment will make the child more comfortable Computerized incentives (e.g., animations, games) can improve effort and engagement Use of a nose clip and posture should always be documented as well as any medications that the child is taking and when they were last taken 3. The Spirometry Maneuver Careful observation during the maneuver is critical. Mouth fully sealed around the mouthpiece to prevent air leaks and if needed, a smaller mouthpiece Curves should be visually inspected for a rapid rise to peak flow, a smooth descending limb on the flow-volume curve, and no coughing or holding of breath. Minimum of three maneuvers (no maximum limit) A Key Adjustment: FEV 0.5 or FEV 0.75 Instead of FEV 1 The ATS/ERS guideline allows for a minimum expiratory time of 3 seconds in children under 10, compared to 6 seconds for adults. Many preschool children cannot sustain forced expiration for more than one second. For this reason, FEV in 0.5 seconds or 0.75 seconds (FEV 0.5 or FEV 0.75 ) is considered an acceptable and clinically useful alternative. (2) Using age-appropriate measures helps prevent underestimating lung function simply because of developmental limitations. In-Clinic vs. At-Home Spirometry With advances in digital health, at-home spirometry has become increasingly common. But how does it compare to clinic-based testing? In-Clinic Spirometry In-clinic testing typically follows all ATS/ERS best practice guidelines, including (2): Real-time technician coaching and familiarization with equipment Immediate quality assessment Spirometers are very precise and must meet certain requirements Strict repeatability criteria The presence of a trained specialist can significantly improve technique and effort, especially in young children. At-Home Spirometry At-home spirometry follows similar principles, but some clinical requirements are difficult—or impossible—to replicate: No operator/technician present so no real-time coaching, limited ability to assess subtle quality errors, and fewer opportunities for immediate correction Many validated portable spirometers do not provide visual flow-volume or volume-time curves (3) Overall, less strict adherence to ATS/ERS technical criteria Because of these differences, home spirometry values are often slightly lower and less consistent than clinic values (7, 8, 3). However, home spirometry has been shown to be feasible and reliable at identifying changes from baseline over time, correlating strongly with clinic-based spirometry (7, 8, 3, 9, 10). If the flow-volume curve appears appropriate and the effort is consistent, slightly lower numbers do not invalidate the test (3). Some portable spirometers, like the Spirobank Smart, have been shown to be valid tools for assessing lung function and can even significantly predict COPD (1). Practical Takeaways Preschool children can perform spirometry with appropriate modifications. FEV 0.5 or FEV 0.75 is acceptable when forced expiration beyond 1 second is not achievable. Strict attention to maneuver quality is critical Home spirometry is valid and reliable—even if values are slightly lower than clinic measurements. Spirometry in preschool children requires patience, adaptation, and thoughtful interpretation. Whether performed in clinic or at home, high-quality spirometry can provide valuable insights into pediatric lung health when best practices are followed, and results are interpreted within the appropriate developmental context. References Lin C-H, Cheng S-L, Wang H-C, Hsu W-H, Lee K-Y, Perng D-W, Lin H-I, Lin M-S, Tsai J-R, Wang C-C, Lin S-H, Wang C-Y, Chen C-Z, Yang T-M, Liu C-L, Wang T-Y, Lin M-C. Novel App-Based Portable Spirometer for the Early Detection of COPD. Diagnostics (Basel) 11: 785, 2021. doi: 10.3390/diagnostics11050785. Beydon N, Davis SD, Lombardi E, Allen JL, Arets HGM, Aurora P, Bisgaard H, Davis GM, Ducharme FM, Eigen H, Gappa M, Gaultier C, Gustafsson PM, Hall GL, Hantos Z, Healy MJR, Jones MH, Klug B, Lødrup Carlsen KC, McKenzie SA, Marchal F, Mayer OH, Merkus PJFM, Morris MG, Oostveen E, Pillow JJ, Seddon PC, Silverman M, Sly PD, Stocks J, Tepper RS, Vilozni D, Wilson NM. An Official American Thoracic Society/European Respiratory Society Statement: Pulmonary Function Testing in Preschool Children. Am J Respir Crit Care Med 175: 1304–1345, 2007. doi: 10.1164/rccm.200605-642ST. Ramos Hernández C, Núñez Fernández M, Pallares Sanmartín A, Mouronte Roibas C, Cerdeira Domínguez L, Botana Rial MI, Blanco Cid N, Fernández Villar A. Validation of the portable Air-Smart Spirometer. PLoS One 13: e0192789, 2018. doi: 10.1371/journal.pone.0192789. Nève V, Edmé J, Devos P, Deschildre A, Thumerelle C, Santos C, Methlin C, Matran M, Matran R. Spirometry in 3–5‐year‐old children with asthma. Pediatric Pulmonology 41: 735–743, 2006. doi: 10.1002/ppul.20389. Moore VC. Spirometry: step by step. Breathe 8: 232–240, 2012. doi: 10.1183/20734735.0021711. Coates AL, Graham BL, McFadden RG, McParland C, Moosa D, Provencher S, Road J. Spirometry in primary care. Can Respir J 20: 13–22, 2013. doi: 10.1155/2013/615281. Oppenheimer J, Hanania NA, Chaudhuri R, Sagara H, Bailes Z, Fowler A, Peachey G, Pizzichini E, Slade D. Clinic vs Home Spirometry for Monitoring Lung Function in Patients With Asthma. CHEST 164: 1087–1096, 2023. doi: 10.1016/j.chest.2023.06.029. Paynter A, Khan U, Heltshe SL, Goss CH, Lechtzin N, Hamblett NM. A comparison of clinic and home spirometry as longtudinal outcomes in cystic fibrosis. J Cyst Fibros 21: 78–83, 2022. doi: 10.1016/j.jcf.2021.08.013. Ramsey RR, Plevinsky JM, Milgrim L, Hommel KA, McDowell KM, Shepard J, Guilbert TW. Feasibility and Preliminary Validity of Mobile Spirometry in Pediatric Asthma. J Allergy Clin Immunol Pract 9: 3821–3823, 2021. doi: 10.1016/j.jaip.2021.06.005. Barker NJ, Kirkby J, Robson EA, Price OJ, Burns PD, Fettes E, Stokes LS, Elphick HE, Sails J. P63 Multicentre prospective cohort study of remote lung function testing in children: validation and comparison of supervised and unsupervised spirometry. Thorax 78: A144–A144, 2023. doi: 10.1136/thorax-2023-BTSabstracts.215.

par Ethan Wilson, M.Sc., B.Sc.
•
4 février 2026
Serious gam es are games designed with a purpose beyond pure entertainment. While they may look and feel like traditional video games, their primary goals are often to teach users new skills, improve knowledge, or encourage behaviour changes that can be transferred into real-world settings (1). Well-designed serious games are challenging and fun, while simultaneously incorporating these features (1). In the context of respiratory diseases, serious games often take a multifaceted approach. They may combine disease education, training of technical skills (such as inhaler use or spirometry), behaviour change strategies, and self-monitoring to support long-term disease management. By embedding these elements into an engaging and interactive experience, serious games aim to make learning both effective and enjoyable. Why Serious Games? Serious games are increasingly being explored in healthcare to address gaps where traditional interventions fall short. This is particularly relevant in asthma care. Even with the standard pharmacological treatments, asthma patients still often have poor symptom control and frequent exacerbations (2). Studies have consistently shown that asthma education improves disease management and reduces emergency department visits and hospitalizations (1). Despite this, attendance and engagement at conventional asthma education programs remains low (1, 2). Children present a unique challenge. In preschool-aged children (2–5 years old), there is a significant gap in normative respiratory data, including reference values for lung function (3, 4) because without the ability to measure, the data is not available. Much of the data currently used in this age group is extrapolated from older children or infants, which may limit accuracy and clinical usefulness (3). Frequent asthma exacerbations are common, and an estimated 40–55% of children with asthma are considered uncontrolled (5, 6). At the same time, self-monitoring of peak expiratory flow (PEF), a lung function measure, can improve medication adherence and decrease exacerbations, but adherence to PEF monitoring is difficult to achieve in pediatric populations (7–10). On the other hand, patients report a positive outlook on the efficiency, convenience, and control of their care with home monitoring (11, 12). Serious games aimed at educating children about asthma and respiratory health may help bridge this gap. By presenting information in a playful, age-appropriate format, these games can motivate children to engage with concepts that might otherwise feel abstract or intimidating. Coupling with respiratory monitoring adds another dimension to self-care in this group, aligning objective metrics with management and education goals. Serious Games and Respiratory Monitoring Regarding respiratory health, serious games are being explored as tools to support respiratory monitoring and skill acquisition. In a study involving children and adults with cystic fibrosis (CF), unsupervised home spirometry was generally viewed as interesting and favourable by users (11). While portable spirometry measures may lack the absolute precision of institutional-clinical-grade spirometry, measures still highly correlate with hospital measurements and patients are willing and able to engage with home-based monitoring, highlighting the potential for improved training tools and feedback mechanisms (11–13). The true value lies in longitudinal tracking, identifying a personal baseline, and detecting clinically relevant intra-patient deviations from that baseline. Adherence to PEF monitoring in pediatric populations is typically low but gaming programs have been seen to improve motivation and increase adherence to exercise programs with promising results in asthma (2). In combination with the increase in medication adherence when self-monitoring PEF, gamifying asthma management—by turning measurements into part of a game or challenge—may help sustain engagement and improve long-term adherence. Some studies have demonstrated that serious games can motivate children to adhere to asthma medications and a pilot study also suggests that these games are acceptable and feasible for pediatric use, although further development and validation are needed (6, 14). Importantly, cooperation during spirometry tasks embedded within serious games has been encouraging, with successful spirometry achieved in approximately 70% of children (3). A review of serious games in asthma education suggested that their effectiveness may be enhanced when delivered as mobile applications (1). Mobile platforms allow for greater accessibility, flexibility, and integration into daily routines. The review also emphasized that serious games should move beyond simple information delivery and instead focus on simulating real-world scenarios or training behaviours that promote meaningful behaviour change (1). Do Serious Games Work? The growing body of evidence suggests that serious games can be effective tools in pediatric respiratory care, particularly when they are thoughtfully designed and tailored to their target audience. SPARKY is a serious game developed to train spirometry in children who are naïve to the procedure, supporting diagnosis and enabling continued monitoring. The goal of SPARKY is to teach children how to perform the spirometry manoeuvres correctly by integrating the technique into an interactive and engaging game environment and then keeping them engaged to support continued monitoring to better manage asthma control and medication adherence. By focusing on skill acquisition through play, SPARKY illustrates how serious games can transform a technically challenging medical task into a child-friendly experience and give healthcare workers and parents monitoring tools to improve outcomes for a previously undertreated age group. Looking Ahead While serious games are not a replacement for traditional medical care, they represent a promising complementary approach—especially in pediatric populations where engagement, motivation, and adherence are ongoing challenges. As mobile technology advances and evidence continues to grow, serious games may play an increasingly important role in respiratory education, monitoring, and self-management. Drummond D , Monnier D , Tesnière A , Hadchouel A . A systematic review of serious games in asthma education. Pediatric Allergy & Immunology 28: 257–265, 2017. doi: 10.1111/pai.12690. Darabseh MZ , Badran R , Alhasan EO , Shurrab AM , Amro AA , Mohmara YA , Al Oweidat K , Awwad S , Ledger SJ , Aburub A . Virtual Reality and Videogaming in Pulmonary Rehabilitation for Asthma: A Systematic Review of Clinical Outcomes and Engagement. J Multidiscip Healthc 18: 2281–2292, 2025. doi: 10.2147/JMDH.S508524. Vilozni D , Barker M , Jellouschek H , Heimann G , Blau H . An Interactive Computer-Animated System (SpiroGame) Facilitates Spirometry in Preschool Children. Am J Respir Crit Care Med 164: 2200–2205, 2001. doi: 10.1164/ajrccm.164.12.2101002. Fuhlbrigge AL , Kitch BT , Paltiel AD , Kuntz KM , Neumann PJ , Dockery DW , Weiss ST . FEV1 is associated with risk of asthma attacks in a pediatric population. Journal of Allergy and Clinical Immunology 107: 61–67, 2001. doi: 10.1067/mai.2001.111590. Sarasmita MA , Larasanty LPF , Kuo L-N , Cheng K-J , Chen H-Y . A Computer-Based Interactive Narrative and a Serious Game for Children With Asthma: Development and Content Validity Analysis. J Med Internet Res 23: e28796, 2021. doi: 10.2196/28796. Silva-Lavigne N , Valderrama A , Pelaez S , Bransi M , Balli F , Gervais Y , Gaudy T , Tse SM . Acceptability of Serious Games in Pediatric Asthma Education and Self-management: Pilot Study. JMIR Pediatr Parent 5: e33389, 2022. doi: 10.2196/33389. Chelabi K , Balli F , Bransi M , Gervais Y , Marthe C , Tse SM . Validation of a Portable Game Controller to Assess Peak Expiratory Flow Against Conventional Spirometry in Children: Cross-sectional Study. JMIR serious games 9: e25052, 2021. doi: 10.2196/25052. Feldman JM , Kutner H , Matte L , Lupkin M , Steinberg D , Sidora-Arcoleo K , Serebrisky D , Warman K . Prediction of peak flow values followed by feedback improves perception of lung function and adherence to inhaled corticosteroids in children with asthma. Thorax 67: 1040–1045, 2012. doi: 10.1136/thoraxjnl-2012-201789. Burkhart PV , Rayens MK , Oakley MG , Abshire DA , Zhang M . Testing an Intervention to Promote Children’s Adherence to Asthma Self-Management. Journal of Nursing Scholarship 39: 133–140, 2007. doi: 10.1111/j.1547-5069.2007.00158.x. Paton JY . Republished: Perception of lung function, adherence to inhaled corticosteroids, and the role of peak expiratory flow feedback in paediatric asthma. Postgrad Med J 89: 61–62, 2013. doi: 10.1136/postgradmedj-2012-202244rep. Davis J , Ryan M , Marchetti P , Dahlberg SE , Greenberg J , Bacon C , Kaur R , Scalia S , Sawicki GS . Real‐world feasibility of short‐term, unsupervised home spirometry in CF. Pediatric Pulmonology 57: 3129–3135, 2022. doi: 10.1002/ppul.26147. Boente RD , Schacht S , Borton R , Vincent J , Golzarri-Arroyo L , Rattray N . Assessing the acceptability and feasibility of remote spirometric monitoring for rural patients with interstitial lung disease: a multimethod approach. Respir Res 25: 92, 2024. doi: 10.1186/s12931-024-02735-z. Moor CC , Wapenaar M , Miedema JR , Geelhoed JJM , Chandoesing PP , Wijsenbeek MS . A home monitoring program including real-time wireless home spirometry in idiopathic pulmonary fibrosis: a pilot study on experiences and barriers. Respir Res 19: 105, 2018. doi: 10.1186/s12931-018-0810-3. Poot CC , de Boer J , Goto L , van de Hei SJ , Chavannes NH , Visch VT , Meijer E . The Design of a Persuasive Game to Motivate People with Asthma in Adherence to Their Maintenance Medication. Patient Prefer Adherence 17: 2719–2736, 2023. doi: 10.2147/PPA.S423161.
par Ethan Wilson, B.Sc., M.Sc.
•
21 janvier 2026
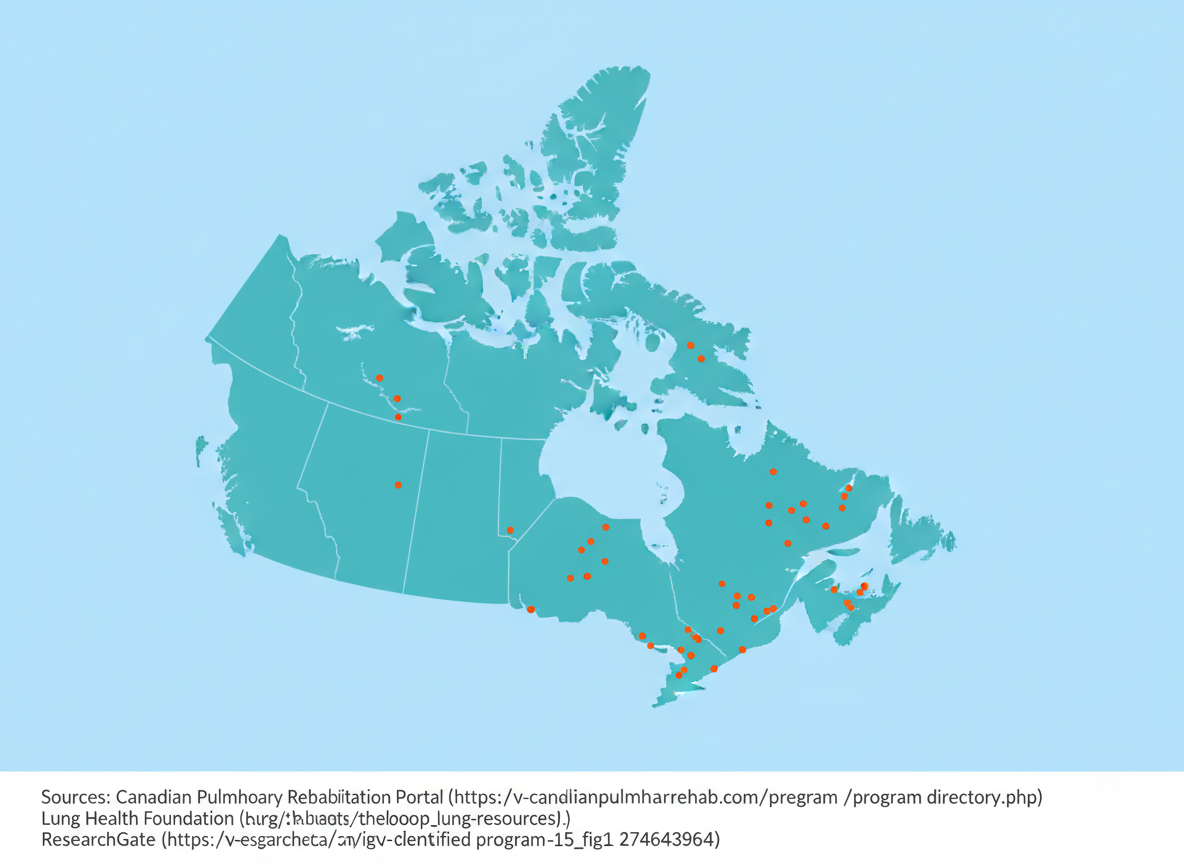
Pulmonary rehabilitation (PR) has long been a cornerstone in managing chronic respiratory diseases. Traditionally used for adults living with chronic obstructive pulmonary disease (COPD), PR programs are now being explored for people with asthma and even for children. These programs have proven benefits—helping patients breathe easier, build endurance, and improve quality of life. But despite the evidence, access to pulmonary rehabilitation remains a major challenge across Canada and the United States. What Is Pulmonary Rehabilitation? Pulmonary rehabilitation combines exercise training, education, and support to help people with chronic lung conditions live better and stay active. A typical program involves physiotherapists, respiratory therapists, and nurses who guide patients through safe exercise plans and breathing strategies. The benefits are clear: PR improves exercise capacity, reduces hospital admissions, and enhances overall well-being. Yet for many patients, simply finding a program nearby is the hardest part. Access to Pulmonary Rehab in Canada Access to PR programs in Canada has been alarmingly low for years. A national survey in 2005 found that only 1.2% of Canadians with COPD had access to pulmonary rehabilitation. At that time, no programs existed in any of the territories, Prince Edward Island, or Newfoundland and Labrador . A decade later, things hadn’t improved much. By 2015, all provinces had at least one PR program—but still none existed in the territories. Most were clustered along the Canada–U.S. border , making them nearly inaccessible for rural residents. Researchers estimated that only 0.4% of Canadians could access a PR program, suggesting that growth in services had not kept up with the rising number of COPD patients (1). For many, travel distance is the biggest barrier. Studies show that travel times over 30 minutes significantly reduce attendance rates (2, 3). Tele-Pulmonary Rehab: A Promising Solution To address these challenges, Canada has started exploring tele-pulmonary rehabilitation —programs delivered via live video conferencing. Researchers in Quebec explored delivering tele-PR by having physiotherapists from central hospitals connect with patients in remote areas through local community centers. Early results are encouraging, with 83% of participants completing the tele-PR programs , compared to 72% for the traditional in-person rehab. Tele-PR also showed higher follow-up rates, suggesting that convenience and accessibility could make a real difference. Further research should look to evaluate home-based tele-PR , which could further bridge the accessibility gap (2). Pulmonary Rehab Access in the United States The U.S. faces similar challenges. While urban residents often have reasonable access, rural communities are left behind. A 2024 study reported that nearly 47.8% of the U.S. population lives within a 15-minute drive of a PR center, and another 32.5% (80.3% total) are within 30 minutes. But in rural regions, some patients face drives of over an hour to reach the nearest program (4). As of 2023, there were 1,696 PR centers across the country —roughly one center for every 6,000 Americans with COPD . Although 61.5% of the population had a center within 10 miles, just 11.3% of rural households had a center within that range—meaning rural residents are 95% less likely to have nearby access compared to those in major cities (5). Closing the Gap Pulmonary rehabilitation works—but it can only help those who can reach it. Both Canada and the United States are seeing clear urban–rural divides in access. Expanding telehealth options, funding community-based programs, and improving transportation support could help close this gap. As research continues to show the effectiveness of both in-person and virtual rehabilitation, it’s time to make sure that every person with chronic lung disease has access—not just those living near major hospitals. Camp PG , Hernandez P , Bourbeau J , Kirkham A , Debigare R , Stickland MK , Goodridge D , Marciniuk DD , Road JD , Bhutani M , Dechman G . Pulmonary Rehabilitation in Canada: A Report from the Canadian Thoracic Society COPD Clinical Assembly. Canadian Respiratory Journal 22: 369851, 2015. doi: 10.1155/2015/369851. Alwakeel AJ , Sicondolfo A , Robitaille C , Bourbeau J , Saad N . The Accessibility, Feasibility, and Safety of a Standardized Community-based Tele-Pulmonary Rehab Program for Chronic Obstructive Pulmonary Disease: A 3-Year Real-World Prospective Study. Annals ATS 19: 39–47, 2022. doi: 10.1513/AnnalsATS.202006-638OC. Sabit R , Griffiths TL , Watkins AJ , Evans W , Bolton CE , Shale DJ , Lewis KE . Predictors of poor attendance at an outpatient pulmonary rehabilitation programme. Respiratory Medicine 102: 819–824, 2008. doi: 10.1016/j.rmed.2008.01.019. Kahn PA , Mathis WS . Accessibility of Pulmonary Rehabilitation in the US. JAMA Netw Open 7: e2354867, 2024. doi: 10.1001/jamanetworkopen.2023.54867. Rochester CL . Insufficient Patient Access to Pulmonary Rehabilitation: A Multifaceted Problem. Ann Am Thorac Soc 20: 510–512, 2023. doi: 10.1513/AnnalsATS.202301-032ED.

par Ethan Wilson, B.Sc., M.Sc.
•
6 janvier 2026
Asthma can make everyday activities — like running, playing, or even laughing a little too hard — a challenge for some children (1). Traditional asthma treatment has long focused on medication, but there’s another piece of the puzzle that’s often overlooked: pulmonary rehabilitation (PR). Despite being the most common chronic respiratory disease in children around the world, pulmonary rehabilitation (PR) has not been well explored as a pediatric treatment tool for asthma (2, 3). Most PR programs have been focused on adults with chronic obstructive respiratory disorder (COPD), but there has recently been more research looking into the benefits of PR in children. Let’s take a look into what it is, how it works, and how it can help your child breathe easier! What Is Pulmonary Rehabilitation? Pulmonary rehabilitation is a multidisciplinary program that combines: Exercise training Education Breathing techniques and breath training Lifestyle and behavioural change Together, these components help people with chronic respiratory conditions improve their health, manage symptoms, and enhance overall quality of life (4–6). PR programs are specially designed to promote changes that are supportive of long-term respiratory health (2). How Pulmonary Rehabilitation Helps Children With Asthma Although PR is more established in adult care, pediatric research is beginning to reveal several promising benefits. Studies have shown that pulmonary rehabilitation and exercise training can lead to improvements in lung function, inspiratory muscle strength, asthma control, and quality of life for children and caregivers alike (1, 2, 7, 8). Some studies have reported that PR can also improve exercise performance although this result is still up for debate (1, 2, 8). Where Does the Research Stand? Overall, the evidence shows that pulmonary rehabilitation is a promising and viable option for improving quality of life and lung function in children with asthma (3, 6). Not all studies show improvements across every measure (1, 2, 8). For example, one study found benefits in inspiratory muscle strength, asthma control, and quality of life, but not in spirometry or exercise capacity (2). PR may need to be tailored specifically for pediatric patients, and more research is needed to determine best practices for children of different ages, asthma severities, and fitness levels. Despite these gaps, PR remains a valuable tool that could complement traditional asthma treatments. The Bottom Line Pulmonary rehabilitation is emerging as a powerful ally in pediatric asthma care. While medication remains essential, PR can support children by improving their lung function and quality of life, boosting confidence during physical activity, and enhancing daily quality of life. As research grows, parents and clinicians alike may begin to see PR as a core part of asthma management — not just for adults, but for children too. Abdelbasset WK , Alsubaie SF , Tantawy SA , Abo Elyazed TI , Kamel DM . Evaluating pulmonary function, aerobic capacity, and pediatric quality of life following a 10-week aerobic exercise training in school-aged asthmatics: a randomized controlled trial. Patient Prefer Adherence 12: 1015–1023, 2018. doi: 10.2147/PPA.S159622. Yang S , Zhang Z , Liu Y , Liu E , Luo Z . The Effects of Combined Respiratory Muscle and Exercise Training in Children with Bronchial Asthma: A Randomised Controlled Study. J Asthma Allergy 16: 293–303, 2023. doi: 10.2147/JAA.S398108. Ma Q , Lu M , Yang Q , Gong F , Zhou L , Xu D . Effects of aerobic exercise-based pulmonary rehabilitation on quality of life in pediatric asthma: A systematic review and meta-analysis. Heart & Lung: The Journal of Acute and Critical Care 69: 11–30, 2025. doi: 10.1016/j.hrtlng.2024.09.005. Spruit MA , Singh SJ , Garvey C , ZuWallack R , Nici L , Rochester C , Hill K , Holland AE , Lareau SC , Man WD-C , Pitta F , Sewell L , Raskin J , Bourbeau J , Crouch R , Franssen FME , Casaburi R , Vercoulen JH , Vogiatzis I , Gosselink R , Clini EM , Effing TW , Maltais F , van der Palen J , Troosters T , Janssen DJA , Collins E , Garcia-Aymerich J , Brooks D , Fahy BF , Puhan MA , Hoogendoorn M , Garrod R , Schols AMWJ , Carlin B , Benzo R , Meek P , Morgan M , Rutten-van Mölken MPMH , Ries AL , Make B , Goldstein RS , Dowson CA , Brozek JL , Donner CF , Wouters EFM . An Official American Thoracic Society/European Respiratory Society Statement: Key Concepts and Advances in Pulmonary Rehabilitation. Am J Respir Crit Care Med 188: e13–e64, 2013. doi: 10.1164/rccm.201309-1634ST. Rochester CL , Alison JA , Carlin B , Jenkins AR , Cox NS , Bauldoff G , Bhatt SP , Bourbeau J , Burtin C , Camp PG , Cascino TM , Dorney Koppel GA , Garvey C , Goldstein R , Harris D , Houchen-Wolloff L , Limberg T , Lindenauer PK , Moy ML , Ryerson CJ , Singh SJ , Steiner M , Tappan RS , Yohannes AM , Holland AE . Pulmonary Rehabilitation for Adults with Chronic Respiratory Disease: An Official American Thoracic Society Clinical Practice Guideline. Am J Respir Crit Care Med 208: e7–e26, 2023. doi: 10.1164/rccm.202306-1066ST. Wu P , Hu Y , Yan X . Progress in Research into the Effectiveness of Pulmonary Rehabilitation in Children with Asthma - A Narrative Review. J Asthma Allergy 17: 743–750, 2024. doi: 10.2147/JAA.S475367. Kirkby S , Rossetti A , Hayes D , Allen E , Sheikh S , Kopp B , Patel A . Benefits of pulmonary rehabilitation in pediatric asthma. Pediatric Pulmonology 53: 1014–1017, 2018. doi: 10.1002/ppul.24041. Bingöl Karakoç G , Yilmaz M , Sur S , Ufuk Altintas D , Sarpel T , Güneter Kendirli S . The effects of daily pulmonary rehabilitation program at home on childhood asthma. Allergol Immunopathol (Madr) 28: 12–14, 2000.

par Ethan Wilson, M.Sc., B.Sc.
•
24 décembre 2025
Asth ma attacks can be stressful for both children and parents. Beyond the immediate symptoms, they can lead to missed school days, emergency room visits, and even hospital stays. What many parents don’t realize is that one asthma attack can increase the risk of future ones — but by knowing the warning signs and tracking your child’s lung health, you can take steps to reduce that risk and prepare for an attack. Children who have Repeat Asthma Attacks Research shows that children who have had an asthma attack are more likely to have another one within the next year — about a 25% higher chance (1, 2). Similarly, the more times a child is hospitalized for asthma, the greater the likelihood of another hospital visit down the line (1). One of the key predictors of future asthma problems is a measure called FEV₁ — or forced expiratory volume in one second. It’s a way to see how much air your child can breathe out quickly after taking a deep breath. (Learn what the spirometry lung function measures mean here ) In one study, researchers found that a 10% drop in FEV₁ over three months increased the risk of another asthma flare-up by 28% in the next three months (1, 3). It also meant poor asthma control was more likely (3). Other studies have shown that when FEV₁ levels are below 80% of what’s expected for a child’s age and size, the risk of another asthma attack increases significantly (4). This risk is even greater for FEV 1 less than 60% of the predicted value (4). In other words, even a single lung function test can help predict asthma risks for months — or even years — ahead (5). Another helpful measure is the mid-ma ximal expiratory flow (MMEF), also known as forced expiratory flow25-75% (FEF25 - 75%) . This number represents the average airflow during the middle 50% of a spirometry test. It gives doctors more detail about how well the smaller airways are working (6). One study found that a low MMEF was linked to a higher number of asthma attacks in the following year , making it another valuable tool for predicting which children might be at greater risk (7). What Parents Can Do 1. Monitor Lung Function at Home with SPARKY TM Keeping track of your child’s lung function can help you spot early signs that asthma may be worsening — before symptoms become serious. SPARKY TM lets you measure important lung function values such as FEV₁ and MMEF at home. This gives you and your child’s doctor valuable information to adjust treatment early and prevent an attack. 2. Watch for Changes in Symptoms If your child’s coughing, wheezing, or shortness of breath seems to be getting worse, even slightly, it’s worth noting. Small changes can signal that their asthma is becoming less controlled (8). Keep on top of changes by using the ACT and TRACK surveys regularly (See our blogs for more information). 3. Stick to the Asthma Action Plan Every child with asthma should have a written plan from their healthcare provider. Make sure you know when to give daily medications, when to use a rescue inhaler, when to step up medications and when to seek emergency care (8, 9). 4. Follow Up Regularly Regular checkups — even when your child feels fine — help doctors track trends in lung function and adjust treatment as needed (8). The Bottom Line Asthma attacks don’t have to come as a surprise. By staying aware of your child’s lung function and understanding the risk factors for flare-ups, you can take proactive steps to keep their asthma under control — and help them breathe easier every day. Sarikloglou E , Fouzas S , Paraskakis E . Prediction of Asthma Exacerbations in Children. Journal of Personalized Medicine 14: 20, 2024. doi: 10.3390/jpm14010020. McIntyre A , Busse WW . Asthma Exacerbations: The Achilles Heel of Asthma Care. Trends Mol Med 28: 1112–1127, 2022. doi: 10.1016/j.molmed.2022.09.001. Fielding S , Pijnenburg M , de Jongste JC , Pike KC , Roberts G , Petsky H , Chang AB , Fritsch M , Frischer T , Szefler S , Gergen P , Vermeulen F , Vael R , Turner S . Change in FEV1 and Feno Measurements as Predictors of Future Asthma Outcomes in Children. Chest 155: 331–341, 2019. doi: 10.1016/j.chest.2018.10.009. Kitch BT , Paltiel AD , Kuntz KM , Dockery DW , Schouten JP , Weiss ST , Fuhlbrigge AL . A Single Measure of FEV1 Is Associated With Risk of Asthma Attacks in Long-term Follow-up. CHEST 126: 1875–1882, 2004. doi: 10.1378/chest.126.6.1875. Fuhlbrigge AL , Kitch BT , Paltiel AD , Kuntz KM , Neumann PJ , Dockery DW , Weiss ST . FEV1 is associated with risk of asthma attacks in a pediatric population. Journal of Allergy and Clinical Immunology 107: 61–67, 2001. doi: 10.1067/mai.2001.111590. Lazova S , Priftis S , Petrova G , Naseva E , Velikova T . MMEF25-75 may predict significant BDR and future risk of exacerbations in asthmatic children with normal baseline FEV1. Int J Physiol Pathophysiol Pharmacol 14: 33–47, 2022. Chereches-Panta P , Marica I , Sas V , Bouari-Coblișan AP , Man SC . The Role of Spirometry and MMEF in Pediatric Asthma Monitoring and Prediction of Exacerbations. Children 12: 1398, 2025. doi: 10.3390/children12101398. Asthma Attacks [Online]. Asthma Canada: [date unknown]. https://asthma.ca/get-help/living-with-asthma/asthma-attacks/ [4 Nov. 2025]. Asthma attack - Diagnosis and treatment - Mayo Clinic [Online]. [date unknown]. https://www.mayoclinic.org/diseases-conditions/asthma-attack/diagnosis-treatment/drc-20354274 [4 Nov. 2025].

par Ethan Wilson, M.Sc., B.Sc.
•
10 décembre 2025
Managing asthma isn’t just about treating symptoms when they appear — it’s about understanding how well your asthma is controlled over time and if your child’s asthma is getting worse. Two powerful tools that help both patients and healthcare providers track asthma control are the Asthma Control Test (ACT) and the Test for Respiratory and Asthma Control in Kids (TRACK) . These short surveys take less than 5 minutes to complete and offer valuable insights that can guide treatment, improve communication, and ultimately help prevent asthma flare-ups. What is the Childhood Asthma Control Test (C-ACT)? The ACT is a quick survey designed for children a ged 4-11 . It includes 3 parent-reported and 4 child-reported questions that cover symptoms, activity limitations, and overall control over the past four weeks (1). Child-reported questions are scored from 0 to 3 while parent-reported questions are scored from 0-5. Higher scores represent better asthma control while a final score ranges between 0 and 27 . A total score of 19 or less is considered to represent inadequately controlled asthma (1, 2). Patients and parents can complete the C-ACT in just a few minutes, either on paper or electronically. Clinicians use the score to assess whether a current treatment plan is effective or if adjustments are needed. The link to the C-ACT survey can be found here . What is the Test for Respiratory and Asthma Control in Kids (TRACK) Survey? For children aged 4 or less , who often can’t describe their symptoms accurately, the TRACK is used. This is a 5-question survey that assesses asthma risk and impairment using answers from parents and caregivers to evaluate asthma control (3). This 5-question tool focuses on: Frequency of respiratory symptoms (like coughing or wheezing) Nighttime awakenings Use of quick-relief medications Activity limitations Episodes requiring oral steroids Each question is scored from 0 to 20, for a total out of 100. With a score of 80 or higher, Asthm a is considered well controlled. A score of b elow 80 means Asthma may not be well controlled, and a healthcare review is advised. TRACK is particularly helpful for young children who are just beginning asthma management or whose symptoms fluctuate with viral infections or seasonal changes. The link to the TRACK survey can be found here . Why These Tools Matter Both ACT and TRACK surveys turn subjective experiences into trackable data . They empower families and patients to play an active role in asthma management and help providers make evidence-based decisions. By regularly completing these assessments—often at every check-up or every few months—you can: Detect worsening control before symptoms become severe Evaluate how well current medications are working Foster better communication between patients, families, and providers Support shared decision-making in care plans Using ACT and TRACK at Home Many clinics offer these surveys during appointments, but they can also be completed at home. Online versions and printable PDFs are widely available through organizations such as the Lung Health Foundation, American Lung Association and Asthma and Allergy Foundation of America . Bringing your completed ACT or TRACK results to your next appointment helps your provider fine-tune your care plan and reduce the risk of asthma attacks. The Bottom Line The ACT and TRACK surveys are more than just questionnaires—they’re powerful tools for proactive asthma care . Whether you’re an adult managing your own symptoms or a parent monitoring your child’s breathing, quick check-ins give you a snapshot of the current situation. Staying consistent with these assessments can help ensure fewer flare-ups, better daily living, and greater confidence in managing asthma long-term. Combined with objective metrics such as spirometry, these tools can make a big difference in understanding and controlling asthma. Bime C , Gerald JK , Wei CY , Holbrook JT , Teague WG , Wise RA , Gerald LB . Measurement characteristics of the childhood Asthma-Control Test and a shortened, child-only version. NPJ Prim Care Respir Med 26: 16075, 2016. doi: 10.1038/npjpcrm.2016.75. Burbank AJ , Atkinson CE , Espaillat AE , Schworer SA , Mills K , Rooney J , Loughlin CE , Phipatanakul W , Hernandez ML . Race-specific spirometry equations may overestimate asthma control in Black children and adolescents. Respiratory Research 24: 203, 2023. doi: 10.1186/s12931-023-02505-3. Murphy KR , Zeiger RS , Kosinski M , Chipps B , Mellon M , Schatz M , Lampl K , Hanlon JT , Ramachandran S . Test for Respiratory and Asthma Control in Kids (TRACK): A caregiver-completed questionnaire for preschool-aged children. Journal of Allergy and Clinical Immunology 123: 833-839.e9, 2009. doi: 10.1016/j.jaci.2009.01.058.

par Ethan Wilson, M.Sc., B.Sc.
•
26 novembre 2025
Asthma is the most prevalent respiratory disease in children, affecting ~10% of children across the world (Global Asthma Report) (1). Therefore, assessing lung function in children is essential—especially for those showing signs of persistent coughing, wheezing, or shortness of breath. But when it comes to preschool-aged children (typically ages 3–5), evaluating their lung function isn't as straightforward as it is for older children and adults. One of the gold-standard tools in respiratory medicine, spirometry , is often unreliable in this age group. This gap poses a real challenge for clinicians and can delay the accurate diagnosis and management of conditions like asthma . Why Spirometry Doesn’t Work Well in Preschoolers Cooperation and Comprehension - Spirometry requires a child to Seal their lips tightly around a mouthpiece Take a deep breath in Blow out as hard and as fast as they can for several seconds This sequence is unnatural and therefore challenging for many preschoolers, who may not fully understand or follow instructions consistently (2, 3). The result? Inaccurate, incomplete, or unreliable data . Poor Reproducibility Even if a young child manages to perform the test once, repeating it with the same effort and technique is a major hurdle. Reproducibility and therefore reliability is often lacking in this age group (3, 4). Lack of Normative Data Even when good-quality spirometry is achieved, there’s limited reference data for children under 6 (2). This makes it hard to interpret whether the results are truly normal or not. What Are the Consequences? The inability to perform reliable spirometry in preschoolers leads to several clinical consequences : Delayed or missed diagnosis : Without objective lung function data, diagnosing asthma or other lung diseases becomes more subjective and error-prone. Over-treatment or under-treatment : Children may be prescribed medications they don't need—or may not receive treatment they urgently require. Poor disease monitoring : Clinicians must rely more heavily on symptoms alone when the child has an appointment, which can be vague or misleading due to recall issues. Moving Forward: Bridging the Gap To improve respiratory care in preschoolers, we need: More research into child-friendly lung function tests Broader access to pulmonary function tests and tools that enable lung function tests in this age group Better training and protocols for performing spirometry in young children More normative data for interpreting results in this age group Final Thoughts The preschool years are a critical window for identifying and managing respiratory issues like asthma. Yet our tools for evaluating lung function aren’t always up to the task. While clinicians do their best with clinical judgment and symptom tracking, the lack of reliable objective testing in this age group remains a significant gap in paediatric respiratory care . Continued innovation, research, and advocacy are key to closing it. 1 Yuan L , Tao J , Wang J , She W , Zou Y , Li R , Ma Y , Sun C , Bi S , Wei S , Chen H , Guo X , Tian H , Xu J , Dong Y , Ma Y , Sun H , Lv W , Shang Z , Jiang Y , Lv H , Zhang M . Global, regional, national burden of asthma from 1990 to 2021, with projections of incidence to 2050: a systematic analysis of the global burden of disease study 2021. eClinicalMedicine 80, 2025. doi: 10.1016/j.eclinm.2024.103051. Fuhlbrigge AL , Kitch BT , Paltiel AD , Kuntz KM , Neumann PJ , Dockery DW , Weiss ST . FEV1 is associated with risk of asthma attacks in a pediatric population. Journal of Allergy and Clinical Immunology 107: 61–67, 2001. doi: 10.1067/mai.2001.111590. Aurora P , Stocks J , Oliver C , Saunders C , Castle R , Chaziparasidis G , Bush A . Quality Control for Spirometry in Preschool Children with and without Lung Disease. Am J Respir Crit Care Med 169: 1152–1159, 2004. doi: 10.1164/rccm.200310-1453OC. Nève V , Edmé J , Devos P , Deschildre A , Thumerelle C , Santos C , Methlin C , Matran M , Matran R . Spirometry in 3–5‐year‐old children with asthma. Pediatric Pulmonology 41: 735–743, 2006. doi: 10.1002/ppul.20389.

par Ethan Wilson, M.Sc., B.Sc.
•
12 novembre 2025
It’s been well-documented that racial and ethnic disparities exist across many areas of healthcare. In recent years, however, the focus has begun to shift—from simply recognizing these disparities to actively addressing them. Nowhere is this more critical than in the tools and practices we use to diagnose and manage disease. One such tool is the pulmonary function test (PFT), a standard test used to assess how well the lungs work (1). For over a decade, major medical organizations—including the American Thoracic Society (ATS)—have recommended using race- and ethnicity-specific reference equations to interpret these tests. The idea was to “normalize” expected lung function based on the patient’s racial or ethnic background (2, 3). But this approach has a major flaw: race is a social construct, not a biological one. By embedding race into the very algorithms we use for diagnosis, we risk reinforcing the very disparities we're trying to dismantle. The Problem with Race-Specific Equations Using race-specific equations in pulmonary testing can unintentionally normalize lower lung function values in certain populations—particularly Black individuals. This can lead to underdiagnosis or delayed treatment of pulmonary conditions like asthma or COPD. In fact, it wasn’t until 2022 that the Global Lung Function Initiative (GLI) released race-neutral average reference equations (GLI-Global and GLI-Other), and it was only in 2023 that the ATS and the European Respiratory Society (ERS) formally recommended transitioning to these updated equations (2). What the Research Shows A study by Rosenfeld and colleagues explored what happens when we stop adjusting pulmonary test results for race. They found that using the GLI-Global equations led to a higher average ppFEV1 (percent predicted forced expiratory volume in one second) in White patients (+4.6%) and a lower ppFEV1 in Black patients (-7.7%) (2). This is significant. The decline seen in Black patients with the race-neutral model suggests that prior race-specific equations may have underestimated the severity of lung disease in this population (2). What was once considered "normal" for Black patients may have, in fact, masked serious illness (2). Similarly, a study by Burbank and colleagues found that applying race-neutral spirometry equations led to more children being classified with uncontrolled asthma (ppFEV1 < 80%)—regardless of whether their asthma control test (ACT) labelled them as controlled (ACT >19) or uncontrolled (ACT ≤19). Among children with controlled asthma (measured by ACT score >19), those who shifted from “controlled” (≥80%) to “uncontrolled” (<80%) lung function also had significantly lower FEV1/FVC ratios, indicating worse airway obstruction (3). What This Means for Health Equity These findings highlight a stark reality: race-specific equations, though well-intended, may perpetuate disparities by obscuring the true burden of disease in certain racial and ethnic groups (2, 3). Shifting to race-neutral reference equations is not just a technical correction—it’s a step toward more equitable healthcare. It allows us to detect disease earlier, treat it more effectively, and ensure that our diagnostic tools do not unintentionally reflect or reinforce systemic biases. The Road Ahead As the medical community continues to reevaluate long-standing practices, it’s vital to center health equity in our decisions. The adoption of race-neutral equations in pulmonary function testing is a powerful example of how evidence-based updates can drive more just and inclusive care. While this transition is still unfolding, it represents meaningful progress in aligning science with social responsibility—and in ensuring that all patients, regardless of race, receive the care they truly need. Lung Function Tests [Online]. American Lung Association: [date unknown]. https://www.lung.org/lung-health-diseases/lung-procedures-and-tests/lung-function-tests [7 Oct. 2025]. Rosenfeld M , Cromwell EA , Schechter MS , Ren C , Flume PA , Szczesniak RD , Morgan WJ , Jain R . The impact of switching to race-neutral reference equations on FEV1 percent predicted among people with cystic fibrosis,. Journal of Cystic Fibrosis 23: 443–449, 2024. doi: 10.1016/j.jcf.2024.03.013. Burbank AJ , Atkinson CE , Espaillat AE , Schworer SA , Mills K , Rooney J , Loughlin CE , Phipatanakul W , Hernandez ML . Race-specific spirometry equations may overestimate asthma control in Black children and adolescents. Respiratory Research 24: 203, 2023. doi: 10.1186/s12931-023-02505-3.

par Ethan Wilson, B.Sc., M.Sc.
•
1 novembre 2025
As a parent, watching your child struggle to breathe is one of the most frightening experiences imaginable. Asthma, a common but serious condition, affects thousands of Canadian children and millions of children around the world —and understanding it is the first step toward managing it with confidence. What Is Asthma? Asthma is a chronic lung condition that causes inflammation and narrowing of the airways, making it harder to breathe. Children with asthma may experience: Coughing Wheezing Chest tightness Shortness of breath These symptoms can be triggered by allergens, viruses, exercise, or even cold air. While asthma has no cure, it can be managed effectively with the right tools and knowledge. How Is Asthma Diagnosed? Even if your child seems to breathe normally most of the time, asthma can still affect their lung function. A spirometry test to measure how well the lungs are working is most commonly used. This test is part of a group called Pulmonary Function Tests (PFTs). Key Measurements in Spirometry: Forced Vital Capacity (FVC): The total amount of air your child can exhale after taking a deep breath. It helps ensure the test is consistent and reliable. Forced Expiratory Volume (FEV): Measures how much air is exhaled in the first 0.5, 0.75 or one second. Healthcare providers use it to assess if there are limitations in air flow which can be associated with outcomes and severity of obstruction. If this number is below 80% of what’s expected for your child’s age, it may indicate airway obstruction. Peak Expiratory Flow (PEF): Shows how fast and forcefully your child can blow air out. It’s a good indicator of effort and lung strength. Forced Expiratory Flow (FEF): Focuses on the middle part of the breath and is more sensitive to show issues in the smaller airways. It is also referred to as Maximum Mid-Expiratory Flow (MMEF). FEV/FVC Ratio: This ratio compares how much air is forcefully exhaled in the first 0.5 (FEV 0.5 ), 0.75 (FEV 0.75 ) or one (FEV 1 ) second compared to the total exhaled. In children, this value is typically larger due to the larger relative size of their large airways compared to their total lung volume. According to the Global Initiative for Obstructive Lung Diseases (GOLD), a ratio lower than 0.70 may be a signal of more serious lung issues and your clinician may want to monitor more closely. Why This Matters for Your Child Asthma is the most common chronic condition in children, and early detection and management are key. Understanding these measurements helps you and your child’s healthcare provider track progress and adjust treatment plans. Making Lung Health Fun with Sparky™ Managing asthma doesn’t have to be scary or boring. That’s why Lung Games International Inc. created Sparky™, a cuddly digital companion that turns breathing exercises into a game. Using a handheld spirometer as a controller, kids help Sparky™ complete fun challenges—while learning proper breathing techniques and tracking their lung health. It’s a playful way to build healthy habits and give parents peace of mind. Want to learn more about Sparky™ or how to support your child’s lung health? Stay tuned for more tips, tools, and stories from families just like yours. References Asthma - What Is Asthma? NHLBI, NIH [Online]. 2024. https://www.nhlbi.nih.gov/health/asthma [7 Oct. 2025]. Asthma - Symptoms and causes [Online]. Mayo Clinic: [date unknown]. https://www.mayoclinic.org/diseases-conditions/asthma/symptoms-causes/syc-20369653 [7 Oct. 2025]. What Is Asthma? [Online]. Cleveland Clinic: [date unknown]. https://my.clevelandclinic.org/health/diseases/6424-asthma [7 Oct. 2025]. Lung Function Tests [Online]. American Lung Association: [date unknown]. https://www.lung.org/lung-health-diseases/lung-procedures-and-tests/lung-function-tests [7 Oct. 2025]. Spirometry [Online]. American Lung Association: [date unknown]. https://www.lung.org/lung-health-diseases/lung-procedures-and-tests/spirometry [7 Oct. 2025]. Pulmonary Function Test: Purpose, Procedure & Results [Online]. Cleveland Clinic: [date unknown]. https://my.clevelandclinic.org/health/diagnostics/17966-pulmonary-function-testing [7 Oct. 2025]. Graham BL , Steenbruggen I , Miller MR , Barjaktarevic IZ , Cooper BG , Hall GL , Hallstrand TS , Kaminsky DA , McCarthy K , McCormack MC , Oropez CE , Rosenfeld M , Stanojevic S , Swanney MP , Thompson BR . Standardization of Spirometry 2019 Update. An Official American Thoracic Society and European Respiratory Society Technical Statement. Am J Respir Crit Care Med 200: e70–e88, 2019. doi: 10.1164/rccm.201908-1590ST. Nève V , Edmé J , Devos P , Deschildre A , Thumerelle C , Santos C , Methlin C , Matran M , Matran R . Spirometry in 3–5‐year‐old children with asthma. Pediatric Pulmonology 41: 735–743, 2006. doi: 10.1002/ppul.20389. Lazova S , Priftis S , Petrova G , Naseva E , Velikova T . MMEF25-75 may predict significant BDR and future risk of exacerbations in asthmatic children with normal baseline FEV1. Int J Physiol Pathophysiol Pharmacol 14: 33–47, 2022. Kwon DS , Choi YJ , Kim TH , Byun MK , Cho JH , Kim HJ , Park HJ . FEF25-75% Values in Patients with Normal Lung Function Can Predict the Development of Chronic Obstructive Pulmonary Disease. Int J Chron Obstruct Pulmon Dis 15: 2913–2921, 2020. doi: 10.2147/COPD.S261732. Jo YS , Kim SK , Park SJ , Um S-J , Park Y-B , Jung KS , Kim DK , Yoo KH . Longitudinal change of FEV1 and inspiratory capacity: clinical implication and relevance to exacerbation risk in patients with COPD. Int J Chron Obstruct Pulmon Dis 14: 361–369, 2019. doi: 10.2147/COPD.S189384. Peak expiratory flow HealthLink BC [Online]. [date unknown]. https://www.healthlinkbc.ca/healthwise/peak-expiratory-flow [2 Oct. 2025]. Aurora P , Stocks J , Oliver C , Saunders C , Castle R , Chaziparasidis G , Bush A . Quality Control for Spirometry in Preschool Children with and without Lung Disease. Am J Respir Crit Care Med 169: 1152–1159, 2004. doi: 10.1164/rccm.200310-1453OC. Torén K , Schiöler L , Lindberg A , Andersson A , Behndig AF , Bergström G , Blomberg A , Caidahl K , Engvall JE , Eriksson MJ , Hamrefors V , Janson C , Kylhammar D , Lindberg E , Lindén A , Malinovschi A , Lennart Persson H , Sandelin M , Eriksson Ström J , Tanash H , Vikgren J , Johan Östgren C , Wollmer P , Sköld CM . The ratio FEV1/FVC and its association to respiratory symptoms—A Swedish general population study. Clin Physiol Funct Imaging 41: 181–191, 2021. doi: 10.1111/cpf.12684.